The Naked Surgeon: The Naked Truth About Nipple Correction
To begin with, many women commonly refer to the area of the breast containing the areola and nipple collectively as…
The Pacific Center for Plastic Surgery blogs offer additional information about the surgical and non-surgical procedures performed by our board-certified surgeons and expert team. Read through our blog posts to learn more about the procedures that may interest you.

To begin with, many women commonly refer to the area of the breast containing the areola and nipple collectively as…

BE REALISTIC Realistic expectations are key to satisfaction with your results. While improvement is always the goal, perfection is often…
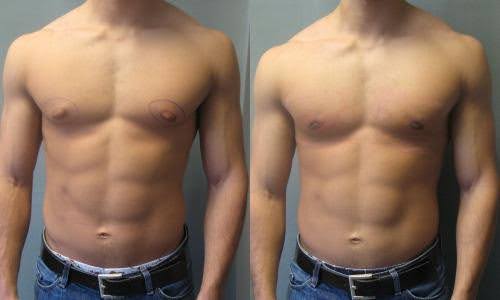
Many men with enlarged male breasts, sometimes referred to as “man boobs,” can feel self-conscious about going shirtless. Gynecomastia is…

If you have loose or sagging skin, lines and wrinkles, or drooping jowls, you may be considering a facelift. It’s…
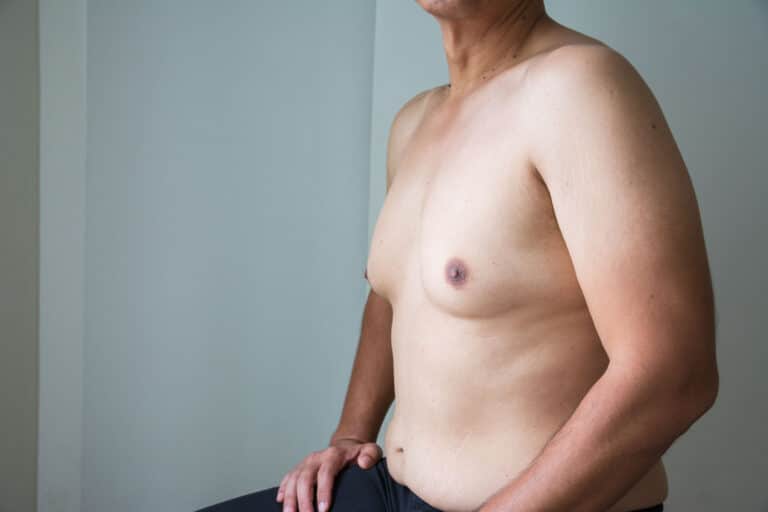
If you look at your chest and hear the unfortunate term “man boobs” in the back of your head, you…

Achieving ideal curves is all about balance. When most of us imagine a sculpted female figure, we imagine a slimmer…


“What can I say about my experience here… It was amazing! Everything I hoped for and more! Not only is Dr. Nichter a perfectionist, he’s also an expert at what he does! I am very pleased with my results! All of his support staff are amazing as well! Everyone makes you feel so welcome and cared for.”*
Laura M. (Google Review)

“The whole process was so easy and I was so well taken care of. 10/10 recommend coming here, don’t even second guess it, you’re at the right place!!! I went to multiple appointments at other surgeons and nothing comes close to how professional and amazing this office is.”*
Dominica F. (Yelp Review)

“Dr. Horowitz is an amazing plastic surgeon who makes everything look so natural and beautiful. He is very kind, artistic and works magic. I am very happy with my results and he did exactly what I wanted/described. I couldn’t be more satisfied. The staff at his office are very patient and supportive throughout the whole process. …”*
Joanne L. (Google Review)

“I had the the most AMAZING experience with Dr. Nichter. I first got breast implants with him back in 2006 and it was perfect. May 2021, I went back to get a full mommy make over with him (breast lift, tummy tuck, liposuction of arms, chin & things). Dr. Nichter and his staffs are amazing from the beginning to end. It has been truly a life changing experience …”*
Patcharin S. (Google Review)

Dr. Horowitz is amazing. I’ve seen him for several surgeries and he is as kind now as he always has been, and knowledgeable too! He is very patient with my millions of questions and works hard to make me feel comfortable and at ease. His staff is great, they are always very welcoming! I love this place. …”*
Irish M. (Google Review)

“Dr. Horowitz is very warm, professional and makes you feel comfortable. He did my breast augmentation for me about a month ago and I couldn’t be happier. My experience was amazing. I didn’t feel rushed during my consultation & he always made sure that I understood everything. …”*
Ashley Z. (RealSelf Review)

“What can I say about my experience here… It was amazing! Everything I hoped for and more! Not only is Dr. Nichter a perfectionist, he’s also an expert at what he does! I am very pleased with my results! All of his support staff are amazing as well! Everyone makes you feel so welcome and cared for.”*
Laura M. (Google Review)

“The whole process was so easy and I was so well taken care of. 10/10 recommend coming here, don’t even second guess it, you’re at the right place!!! I went to multiple appointments at other surgeons and nothing comes close to how professional and amazing this office is.”*
Dominica F. (Yelp Review)

“Dr. Horowitz is an amazing plastic surgeon who makes everything look so natural and beautiful. He is very kind, artistic and works magic. I am very happy with my results and he did exactly what I wanted/described. I couldn’t be more satisfied. The staff at his office are very patient and supportive throughout the whole process. …”*
Joanne L. (Google Review)

Dr. Horowitz is amazing. I’ve seen him for several surgeries and he is as kind now as he always has been, and knowledgeable too! He is very patient with my millions of questions and works hard to make me feel comfortable and at ease. His staff is great, they are always very welcoming! I love this place. …”*
Irish M. (Google Review)

“I had the the most AMAZING experience with Dr. Nichter. I first got breast implants with him back in 2006 and it was perfect. May 2021, I went back to get a full mommy make over with him (breast lift, tummy tuck, liposuction of arms, chin & things). Dr. Nichter and his staffs are amazing from the beginning to end. It has been truly a life changing experience …”*
Patcharin S. (Google Review)

“Dr. Horowitz is very warm, professional and makes you feel comfortable. He did my breast augmentation for me about a month ago and I couldn’t be happier. My experience was amazing. I didn’t feel rushed during my consultation & he always made sure that I understood everything. …”*
Ashley Z. (RealSelf Review)


“What can I say about my experience here… It was amazing! Everything I hoped for and more! Not only is Dr. Nichter a perfectionist, he’s also an expert at what he does! I am very pleased with my results! All of his support staff are amazing as well! Everyone makes you feel so welcome and cared for.”*
Laura M. (Google Review)

“The whole process was so easy and I was so well taken care of. 10/10 recommend coming here, don’t even second guess it, you’re at the right place!!! I went to multiple appointments at other surgeons and nothing comes close to how professional and amazing this office is.”*
Dominica F. (Yelp Review)

“Dr. Horowitz is very warm, professional and makes you feel comfortable. He did my breast augmentation for me about a month ago and I couldn’t be happier. My experience was amazing. I didn’t feel rushed during my consultation & he always made sure that I understood everything. …”*
Ashley Z. (RealSelf Review)

“Dr. Horowitz is an amazing plastic surgeon who makes everything look so natural and beautiful. He is very kind, artistic and works magic. I am very happy with my results and he did exactly what I wanted/described. I couldn’t be more satisfied. The staff at his office are very patient and supportive throughout the whole process. …”*
Joanne L. (Google Review)

Dr. Horowitz is amazing. I’ve seen him for several surgeries and he is as kind now as he always has been, and knowledgeable too! He is very patient with my millions of questions and works hard to make me feel comfortable and at ease. His staff is great, they are always very welcoming! I love this place. …”*
Irish M. (Google Review)

“I had the the most AMAZING experience with Dr. Nichter. I first got breast implants with him back in 2006 and it was perfect. May 2021, I went back to get a full mommy make over with him (breast lift, tummy tuck, liposuction of arms, chin & things). Dr. Nichter and his staffs are amazing from the beginning to end. It has been truly a life changing experience …”*
Patcharin S. (Google Review)

Become a part of BioSpa’s new loyalty program – Ageless. Take control of your appearance and show off your youthful and empowered self with year-long savings on your favorite treatments and products.

Become a part of BioSpa’s new loyalty program – Ageless. Take control of your appearance and show off your youthful and empowered self with year-long savings on your favorite treatments and products.
Connect with Us
(Request a Consultation)
Ready to reach your cosmetic goals with a personalized treatment plan? Fill out our consultation request form to meet with one of our expert team members and find the right treatment for you.
Ready to reach your cosmetic goals with a personalized treatment plan? Fill out our consultation request form to meet with one of our expert team members and find the right treatment for you.